
GxP Lifeline
Revolutionizing Life Sciences Quality Management: Achieve Closed-Loop Quality With Modern Tools

The notion that quality is the sole responsibility of the quality department is outdated. In modern life sciences manufacturing environments, the actions taken by anyone during any stage of the value chain can have an impact on quality – for better or for worse – as a product progresses through each phase of its life cycle. That means quality is the obligation of every employee across the value chain. Anyone involved in any step along the life cycle of a product is a stakeholder in quality.
Keeping people, data, and processes connected across the value chain is challenging, yet it’s imperative for success. With the right set of modern tools, truly closed-loop quality management is attainable.
Digital Quality Management: Unifying the Life Sciences Value Chain
Manufacturers that have implemented modern electronic quality management system (eQMS) software have found it easier to establish an effective closed-loop system that enhances their ability to control costs, risk, and compliance efforts across the value chain.
A modern, robust eQMS provides a single platform for connected quality data, enabling manufacturers to close quality loops in essential functions such as:
- Operations: Closing operational loops requires a centralized system that is accessible across the enterprise. An eQMS expressly designed to close quality loops will prevent vital data from being scattered throughout multiple siloed systems. Since an eQMS enhances data connectivity, it also makes up-to-date information more accessible and simplifies efforts to identify root causes of issues.
- Design: To close quality loops in product design, you must have a system capable of feeding information back to R&D and engineering from all other points along the value chain. A modern eQMS solution provides such capabilities and establishes a digital quality management tool that promotes continuous improvements on future product iterations.
- Supplier Management: Many life sciences manufacturers that rely on suppliers do not have an efficient process for sharing or collaborating on supplier-related information. An eQMS with integrated supplier management software closes quality loops by facilitating collaboration and formalizing the monitoring of supplier quality, designation of responsibilities, etc.
- Service: This final phase of the value chain is the most critical area for establishing closed-loop quality management. Continuous improvement can’t be upheld unless data can be pushed back upstream from this node on the value chain – a feat that’s much easier to accomplish using purpose-built digital quality management tools.
These are just a few examples of the benefits of closed-loop quality management. But if there are so many upsides to closing quality loops with modern tools, why are some companies’ digital quality management initiatives still lagging?
Breaking Down Barriers to Closed-Loop Quality in Life Sciences Manufacturing
Of the 900-plus manufacturers recently surveyed by LNS Research, 78% of them reported that they’re operating “in a state of quality management disconnect.”1 What’s the main contributor to this epidemic of disconnected quality? According to the life sciences professionals who participated in MasterControl’s 2022 quality maturity research study, it’s manual, paper-based processes, which 32% of those surveyed cited as a significant quality pain point.2
Other related issues that cause quality disconnects include technological limitations, lack of communication across the enterprise, reliance on homegrown solutions, and shortsighted IT strategies. A closed-loop quality management system can eliminate all these quality obstacles and limitations by establishing feedback loops that allow performance and operations data to flow bi-directionally.
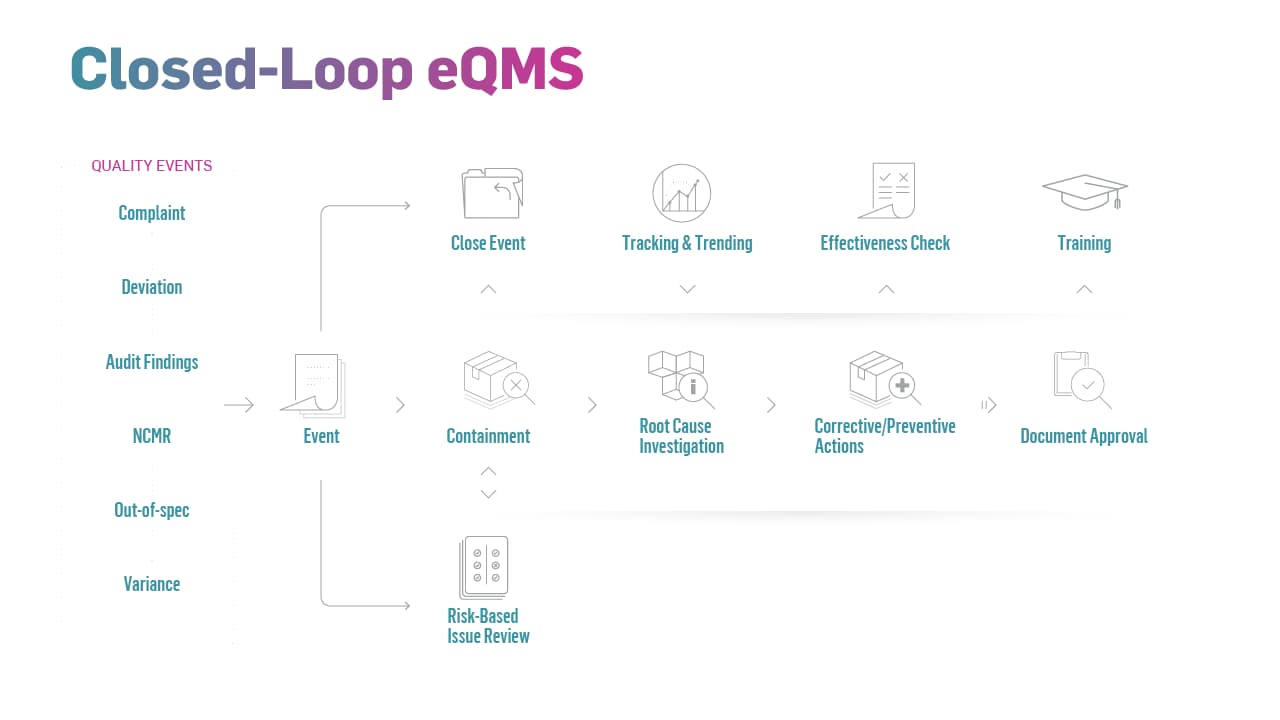
The effectiveness of closed-loop, digital quality management systems is evident, and financial considerations and competitive pressures are driving more manufacturers to adopt them. Ten years ago, fewer than a quarter of the manufacturers reported having closed-loop quality processes.3 Today’s life sciences manufacturers are making greater headway, according to MasterControl’s digital maturity research, which shows the implementation rates of closed-loop, fully digital eQMS solutions are rising, specifically:
- 7% are in the process of implementing an eQMS.
- 33% have partially implemented an eQMS.
- 31% have fully implemented an eQMS at some sites.
- 29% have fully implemented an eQMS at all their sites.4
This progress indicates that companies are at least recognizing the value of digital quality management tools in a broader context and are making strides to enhance the connectivity of quality data across the value chain.
The Rise of Closed-Loop Quality Management Systems in Life Sciences
While most of the LNS Research study respondents who had implemented an eQMS reported their primary goals for doing so was to improve document control and the management of nonconformances, and corrective actions/preventive actions (CAPAs), they said their eQMS solutions have also streamlined other key quality processes such as employee training and complaint handling. In addition, the study noted that a great deal of value is left untapped when companies do not take advantage of eQMS functionalities in deeply interconnected quality processes such as audit, risk, and compliance management.5
For a closed-loop quality management system to be truly effective it must centralize, standardize, and optimize end-to-end business processes and quality data. And nothing has greater impact on quality effectiveness than an eQMS that unites people, processes, and technology across the value chain by serving as a common platform for cross-functional communication, collaboration, and data transfer. And if the eQMS is integration-ready, it can be a force multiplier that takes connected systems beyond their individual capabilities by forging collaboration portals between functional business units and optimizing the delivery of performance data.
Maximizing the Impact of Closed-Loop Quality Management Across Life Sciences Processes
Shrewd executives invest in people, process, and technology across the value chain, but that isn’t enough in today’s highly competitive, fast-paced life sciences industry. Integration and data connectivity are the new keys to success. The integration of enterprise software systems and data sources via a modern eQMS is becoming increasingly more common throughout the industry because it results in demonstrable efficiencies and competitive advantages. A closed-loop eQMS solution can be the hub that unites your product data, diverse teams, and disconnected enterprise systems.
Visit MasterControl’s digital quality management page to discover how a modern eQMS can play a vital role in decreasing risk, reducing your overall costs, and getting your products to market sooner.
References:
- "A Road Map for Addressing Quality and Manufacturing Challenges in Life Sciences,” LNS Research analyst report.
- "The State of Digital Quality Maturity in Pharma and Medtech,” MasterControl research report, 2023.
- Supra note 1.
- Supra note 2.
- Supra note 1.

